Difference between revisions of "Dissolved phosphorus in stormwater runoff - sources and management strategies"
m |
m (→Definitions) |
||
| Line 17: | Line 17: | ||
*[http://bcn.boulder.co.us/basin/data/BACT/info/TP.html City of Boulder, Colorado] | *[http://bcn.boulder.co.us/basin/data/BACT/info/TP.html City of Boulder, Colorado] | ||
*[https://www.water-research.net/index.php/phosphate-in-water Water Research Center] | *[https://www.water-research.net/index.php/phosphate-in-water Water Research Center] | ||
| + | |||
| + | ==Bioavailability of different forms of phosphorus== | ||
| + | Dissolved phosphorus is considered to be more bioavailable than particulate forms of phosphorus. Below is a summary of some studies on bioavailability of phosphorus. | ||
| + | *About 95% of dissolved phosphorus transported to Lake Erie is bioavailable to algae, while only about 30% of the particulate phosphorus attached to eroded sediment is bioavailable ([http://lakeeriealgae.com/different-types-of-phosphorus/ Lake Erie Algae]). | ||
| + | *[https://www.sciencedirect.com/science/article/pii/S0043135406000352 Ellison and Brett] (2006) found on average only 20% of the particulate phosphorus transported in runoff from urban settings was biologically available. | ||
| + | *[https://www.tandfonline.com/doi/full/10.1080/00288330.2013.792851 Abell and Hamilton] (2012) found that about 25% of particulate phosphorus in a stream dominated by stormwater runoff was bioavailable. | ||
| + | *[https://onlinelibrary.wiley.com/doi/full/10.1111/1752-1688.12366 Prestigiacomo et al.] found 10-20% of particulate phosphorus was bioavailable, compared to more than 90% of dissolved phosphorus being bioavailable. Bioavailable phosphorus in the particulate fraction increased somewhat with time after sampling, but never exceeded 30%. | ||
| + | *[https://www.semanticscholar.org/paper/Contribution-of-particulate-phosphorus-to-runoff-Uusitalo-Turtola/54189c9219e05e1b9c66b35a5af7799dc4d4e9a8 Uusitalo et al.] (2003) found 6-10% of particulate phosphorus was bioavailable, but that 34-56% was redox-sensitive, meaning it could become bioavailable under anoxic (reducing) conditions. Other papers corroborate these findings, indicating that a significant portion of particulate phosphorus can become bioavailable under anoxic conditions ([https://www.ncbi.nlm.nih.gov/pubmed/21235180], [https://www.researchgate.net/profile/Colin_Reynolds/publication/229477072_Phosphorus_recycling_in_lakes_Evidence_from_large_limnetic_enclosures_for_the_importance_of_shallow_sediments/links/5a1fdef2458515a4c3d4e69b/Phosphorus-recycling-in-lakes-Evidence-from-large-limnetic-enclosures-for-the-importance-of-shallow-sediments.pdf], [https://www.biogeosciences.net/14/3585/2017/bg-14-3585-2017.pdf], [https://link.springer.com/article/10.1007/BF00024902]) | ||
| + | |||
| + | ==Sources of dissolved phosphorus in runoff== | ||
Revision as of 13:15, 1 October 2021
This page is in development
This page provides a discussion of dissolved phosphorus in stormwater runoff, its sources, and strategies for managing dissolved phosphorus. While the focus is on urban runoff, the basic principles are applicable to agricultural runoff.
Definitions
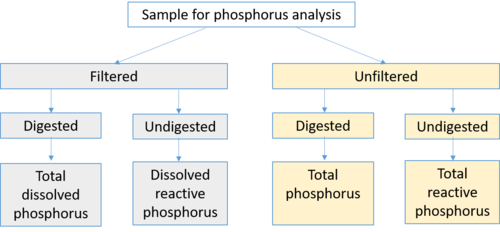
Phosphorus in water is often classified as dissolved (soluble) or particulate (attached to or a component of particulate matter) phosphorus. This nomenclature is somewhat ambiguous, however, as dissolved phosphorus consists of multiple forms of phosphorus, including phosphorus attached to other materials.
- Dissolved phosphorus is typically identified as phosphorus passing through a 0.45 micron filter. It is this dissolved fraction that is considered to be most bioavailable and most difficult to treat.
- Reactive phosphorus is the phosphorus associated with the test for orthophosphate. It consists mostly of orthophosphate but includes a small fraction of other forms.
- Soluble reactive phosphorus is a measure of orthophosphate, the filterable (soluble, inorganic) fraction of phosphorus, the form directly taken up by plant cells.
- Bioavailable phosphorus is the sum of immediately available phosphorus, which can be transformed into an available form by naturally occurring processes.
References for phosphorus forms and testing includes the following.
Bioavailability of different forms of phosphorus
Dissolved phosphorus is considered to be more bioavailable than particulate forms of phosphorus. Below is a summary of some studies on bioavailability of phosphorus.
- About 95% of dissolved phosphorus transported to Lake Erie is bioavailable to algae, while only about 30% of the particulate phosphorus attached to eroded sediment is bioavailable (Lake Erie Algae).
- Ellison and Brett (2006) found on average only 20% of the particulate phosphorus transported in runoff from urban settings was biologically available.
- Abell and Hamilton (2012) found that about 25% of particulate phosphorus in a stream dominated by stormwater runoff was bioavailable.
- Prestigiacomo et al. found 10-20% of particulate phosphorus was bioavailable, compared to more than 90% of dissolved phosphorus being bioavailable. Bioavailable phosphorus in the particulate fraction increased somewhat with time after sampling, but never exceeded 30%.
- Uusitalo et al. (2003) found 6-10% of particulate phosphorus was bioavailable, but that 34-56% was redox-sensitive, meaning it could become bioavailable under anoxic (reducing) conditions. Other papers corroborate these findings, indicating that a significant portion of particulate phosphorus can become bioavailable under anoxic conditions ([1], [2], [3], [4])